Cells naturally produce reactive oxygen species (ROS), but their role in cytoskeletal remodeling has been unclear. In this project I discovered that ROS can serve as a tunable signal for actin dynamics: at intermediate doses they increase filament turnover and accelerate network assembly, while higher doses lead to network disassembly. By using fluorophores as light-activated ROS sources, this behavior can be controlled with high spatial precision. In reconstituted systems, I used this approach to “3D-print” actin structures by selectively turning assembly and disassembly on and off with light, providing a minimal and programmable route to shape cytoskeletal materials in space and time.
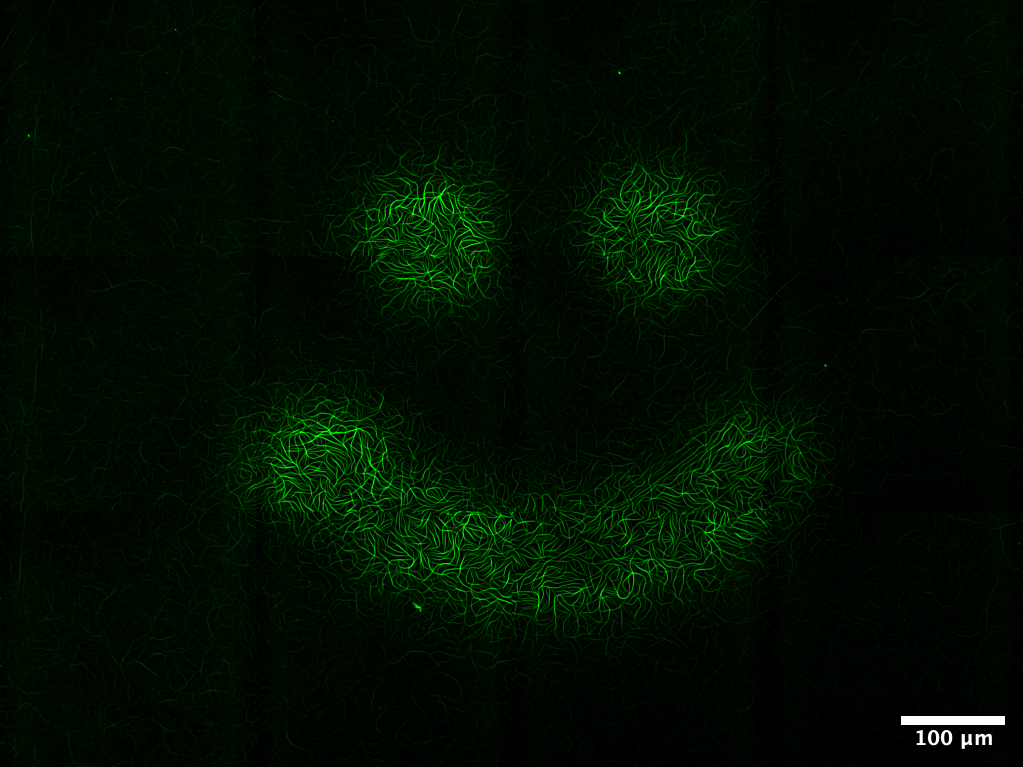
The same mechanism appears to matter in cells. In mammalian cells, localized light-induced ROS generation produces a robust accumulation of actin near the illuminated region, consistent with the growth-promoting regime observed in vitro. In parallel, during wound healing in the jellyfish Clytia hemisphaerica, pharmacological manipulation of ROS alters actin organization at wound edges. Together these results bridge purified systems, cultured cells, and whole tissues, and suggest that ROS can act as a physiological control signal for cytoskeletal remodeling, rather than only as a damaging byproduct of stress.
Litschel T#, Dimitrios Vavylonis, David Weitz (#corresponding author)
“3D printing cytoskeletal networks: ROS-induced filament severing leads to surge in actin polymerization” (Research Paper)
bioRxiv, 2025, DOI: 10.1101/2025.03.19.644260 [Link]
Under Review [Link] at Matter